Your Enterprise-Grade Platform
You want to scale your network and revenue, but ensuring consistency across multiple sites is a logistical nightmare. Luckily, there’s a proven clinical research software platform that easily standardizes data collection and operations. It’s clearly CRIO.
-
40% fewer protocol deviations
-
70% lower audit risk
-
40% higher enrollment
Intuitive Software Solutions Enabling Consistent, Efficient Operations
A powerfully simple clinical research software platform, CRIO has the features you need to take your site network to the next level.
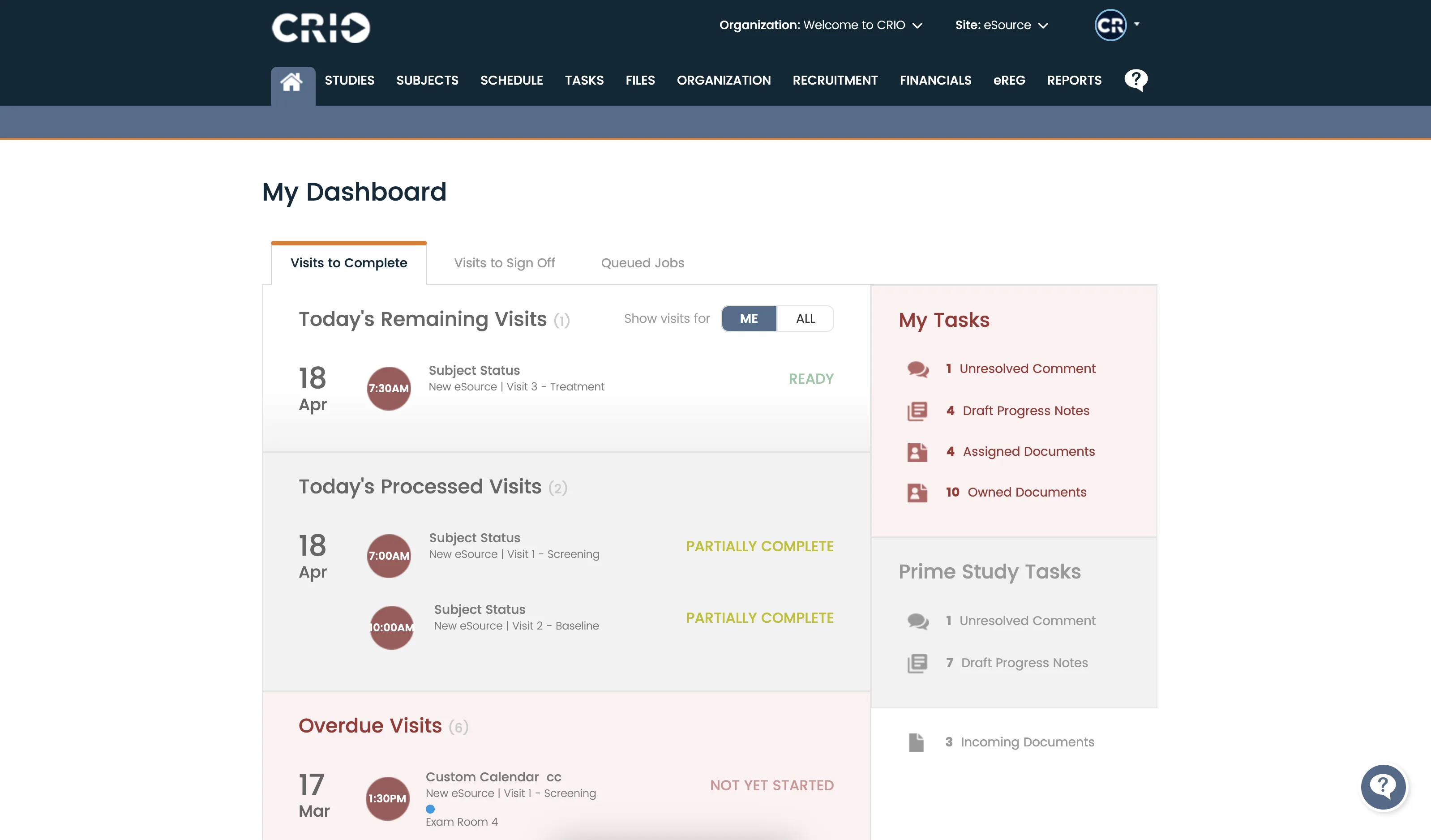
Save Time and Money While Enhancing Quality
- Centralize patient recruitment, study design, regulatory compliance, financial management, and electronic data capture (EDC) entry
- Give your coordinator teams more time to focus on patient recruitment and retention
- Use our open application programming interface (API) to send in recruiting leads, schedule directly on site calendars, and integrate other solutions
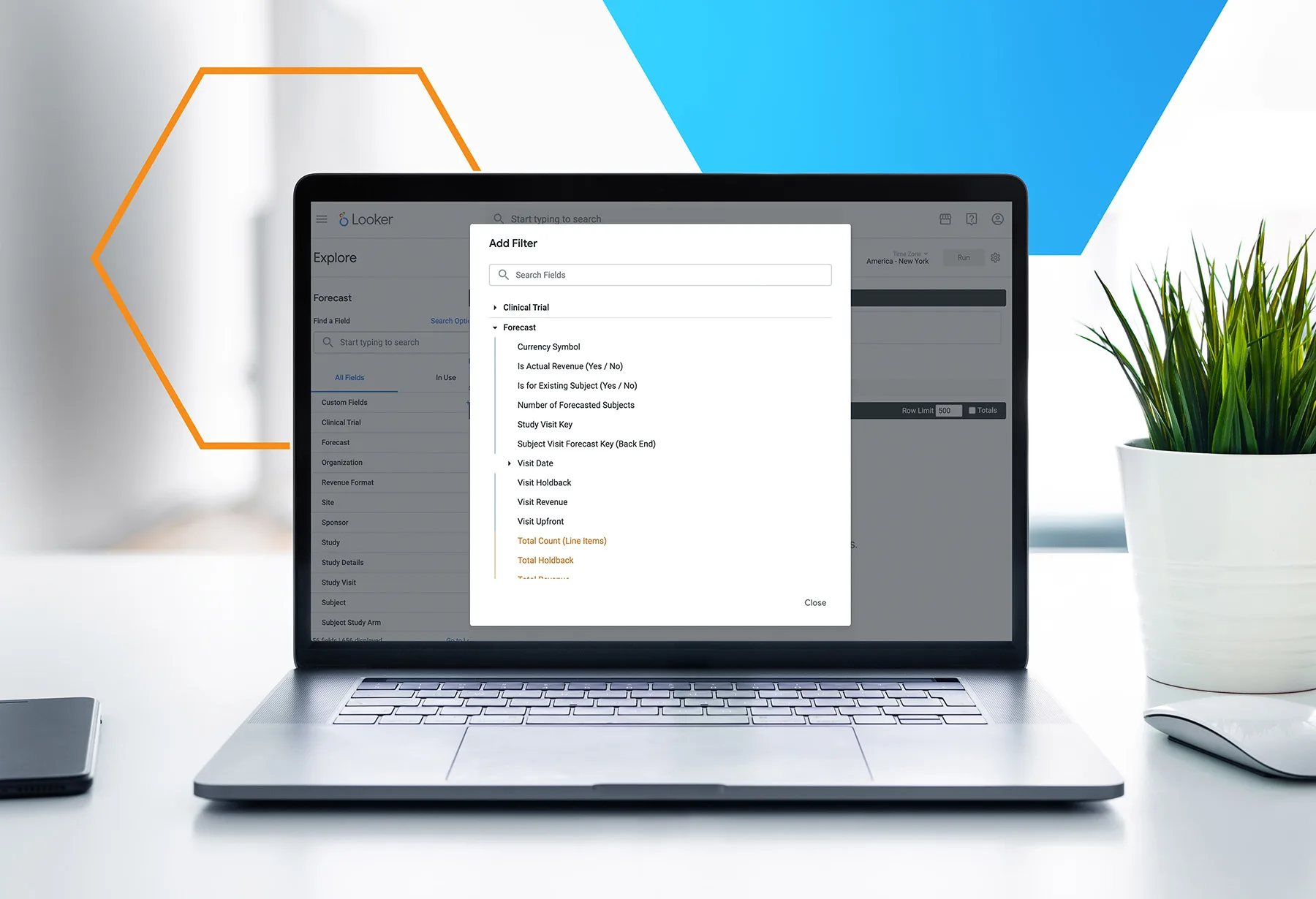
- Leverage our custom reporting and other data access options to get real-time insights for continuous improvement and decision-making
Enhance Reporting
- Design and run hundreds of reports
- Use your own business intelligence tool to query your data directly
eSOURCE-TRIGGERED REVENUE CAPTURE WITH TRANSACTION-LEVEL REPORTING
- Capture all your revenue, costs, and patient stipend obligations in real time, as your team completes eSource, by integrating budgets directly into the eSource template
- Track all changes in revenue at the change item level, enabling consistent monthly revenue closing consistent with GAAP reporting obligations
Get Started Quickly and Keep Going Easily
- Empower staff at all levels with an interactive, AI-powered CRIO help center
- Get streamlined implementation and data migration support from a single point of contact in our dedicated customer experience team
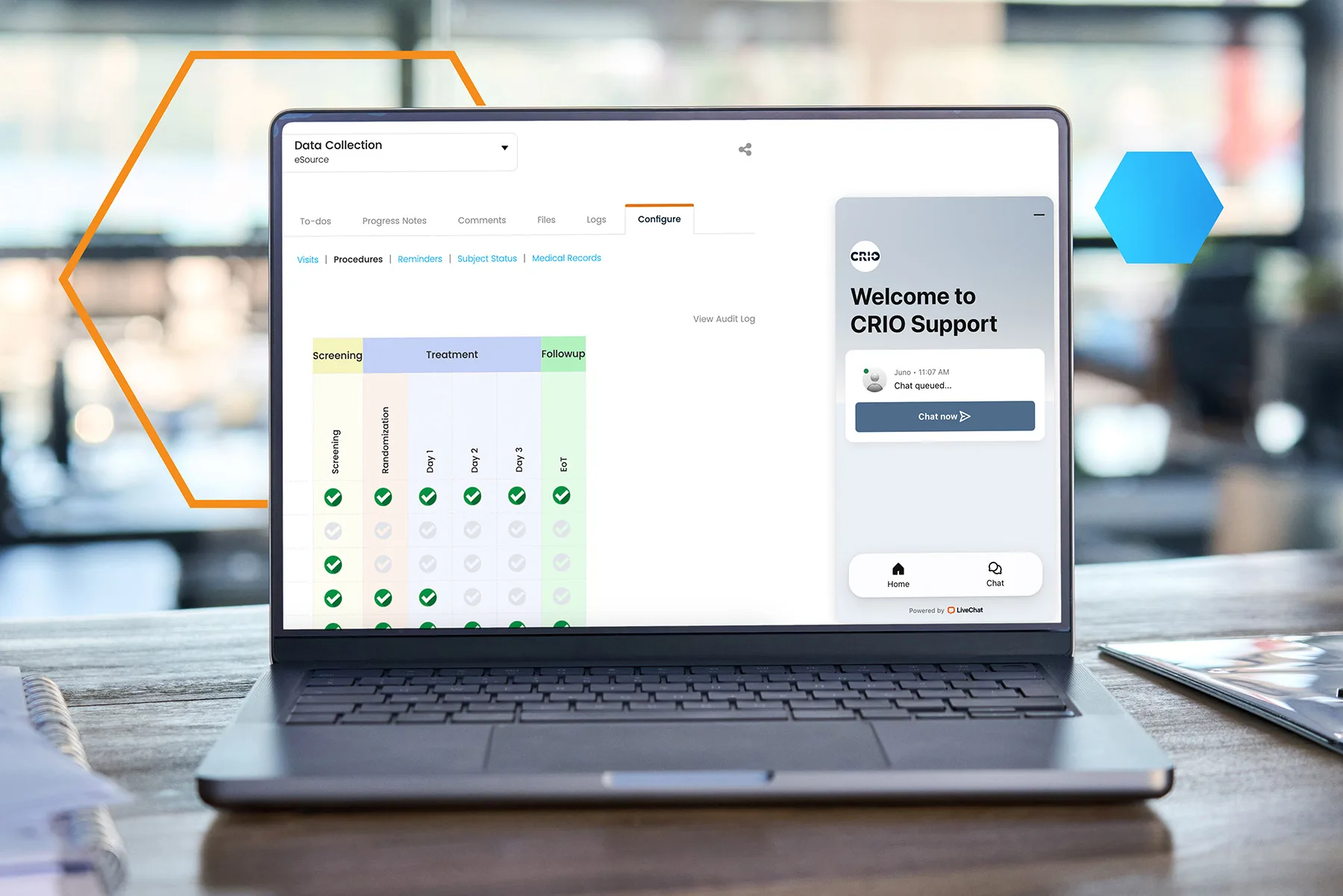
CRIO’s live, 24/7 customer support chat has a 97% satisfaction rating.
See How CRIO Powers the World’s Leading Vaccine Network
Is it possible to reduce data errors, enhance patient safety, and simplify PI oversight on thousands of studies across 9 different sites? With CRIO, it’s not just possible — it’s easy.
Find the Best Clinical Research Software Tools for Your Site Network

How to Simplify Site Network Consolidation With eSource Platforms
We literally wrote the book on using clinical research software to enhance site network consolidation.
The Consolidation & Reinvention of Site Networks