An All-in-One, Intuitive Site Technology
You want to take your site to the next level, but duplicative work and administrative minutia steal focus and overwhelm resources. There’s an easy-to-use clinical research software solution that streamlines workflows to optimize your performance. It’s clearly CRIO.
-
33% time-savings
-
40% fewer protocol deviations
-
70% lower audit risk
Intuitive Software Enabling Better Performance With Less Effort
CRIO is your connected clinical research software solution, offering tight integration between eSource, CTMS, eRegulatory, and eConsent products to reduce duplicative efforts and maximize productivity.
Save Time and Money
- Build study templates in hours — not days — or purchase a template made by our experienced team of study designers and former coordinators
- Eliminate paper binders and enhance PI oversight
- Reduce errors with automated protocol checks
- Access source and regulatory binders in one system, collaborate with your team, and tie electronic delegation of authority (eDOA) duties to source collection
- Never miss a reconsent
- Troubleshoot with live, 24/7 customer service
- Secure sponsor reimbursement for CRIO fees
Grow Your Site
- Manage calling campaigns, send text blasts, build interactive prescreening questionnaires, and pull web leads directly into the system
- Schedule visits and preprogram appointment reminders to keep patient visits on track
Maximize Cash Flow
- Tie finances directly to eSource data
- Keep receivables up to date and manage invoices and payments
- Pay patients through reloadable stipend cards with the click of a button
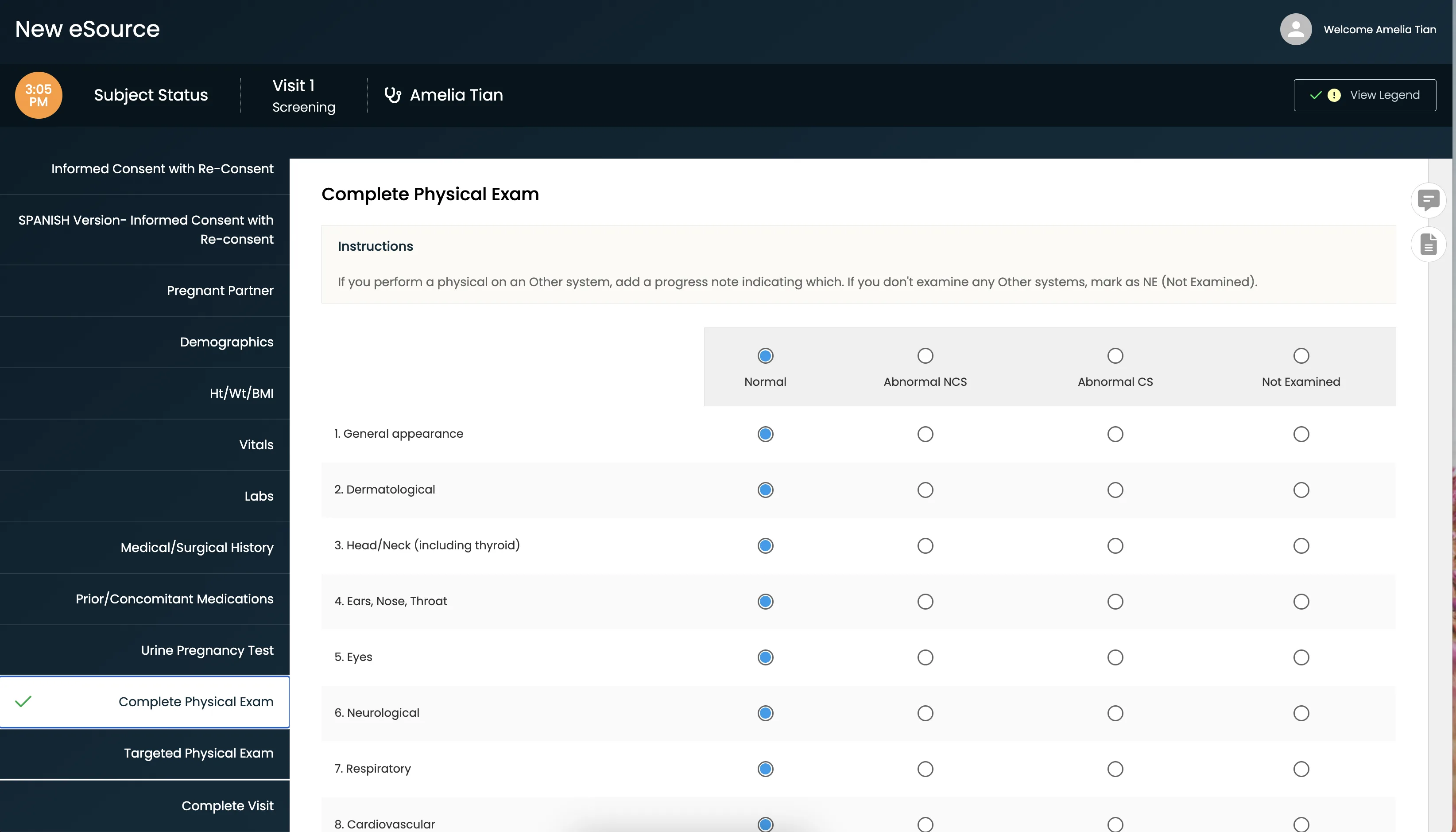
Get a Closer Look at How This Site Saved 33% of Their Time
Thanks to CRIO, clinical research coordinators can screen patients and manage peak patient recruiting times more efficiently — saving time and effort.



